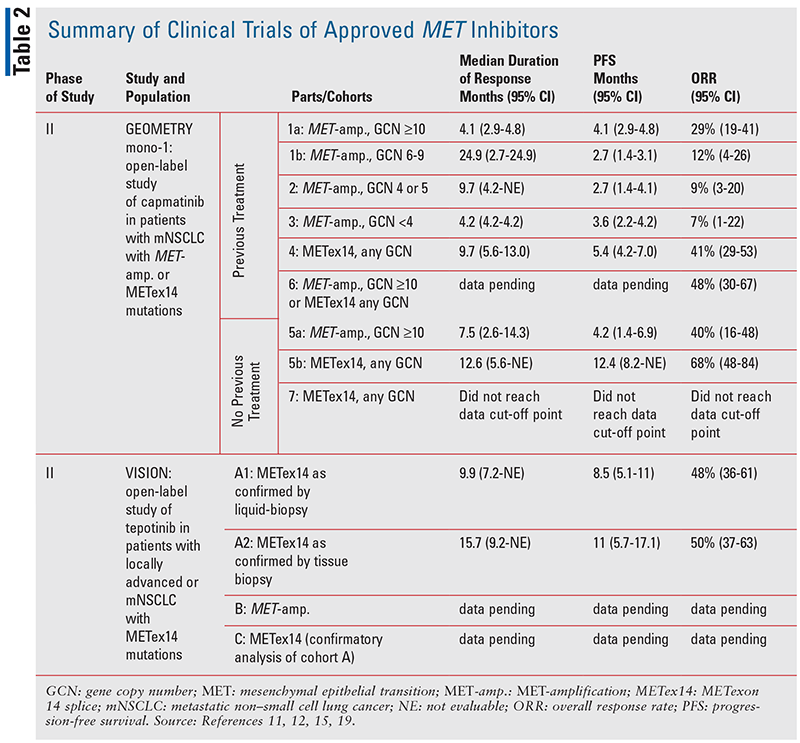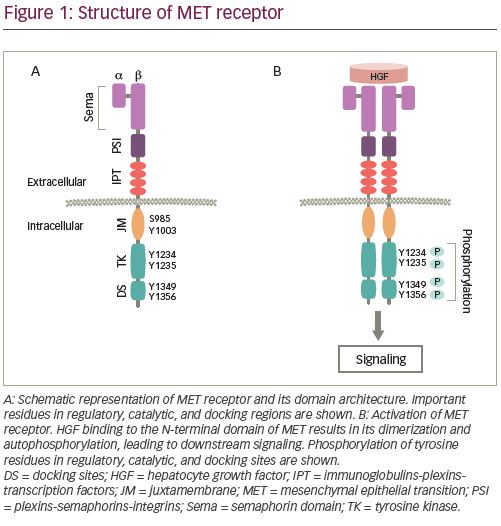
MET Exon 14 Skipping Alterations in NSCLC: Current Understanding and Therapeutic Advances - touchONCOLOGY

Biomedicines | Free Full-Text | Recent Progress and Advances in HGF/MET-Targeted Therapeutic Agents for Cancer Treatment | HTML
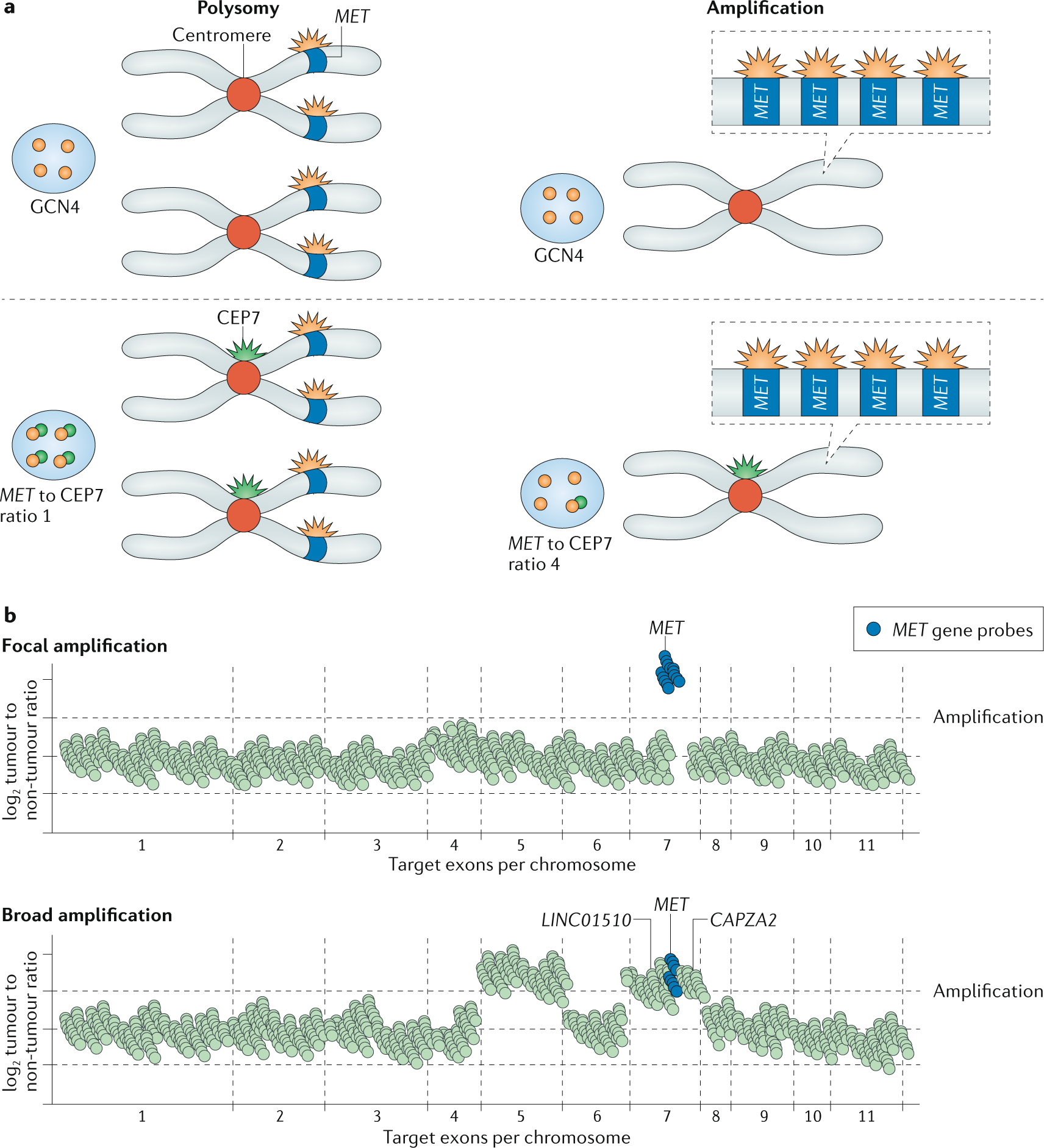
MET-dependent solid tumours — molecular diagnosis and targeted therapy | Nature Reviews Clinical Oncology

Novartis beats Merck KGaA to U.S. finish line with targeted lung cancer drug Tabrecta | Fierce Pharma
Novartis announces MET inhibitor Tabrecta™ approved in Japan for advanced non-small cell lung cancer with METex14 | Novartis

Safety of Tepotinib in Patients with MET Exon 14 Skipping NSCLC and Recommendations for Management - Clinical Lung Cancer

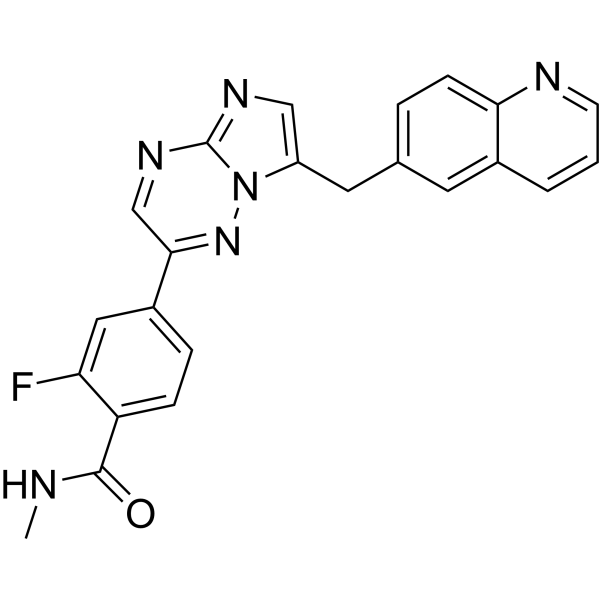
Capmatinib for patients with non-small cell lung cancer with MET exon 14 skipping mutations: A review of preclinical and clinical studies - Cancer Treatment Reviews

Novartis' Powerful MET Inhibitor Tabrecta Has A Strong Therapeutic Effect On METex14 Mutant Non-small Cell Lung Cancer (NSCLC)! - Industry news - News - Hefei Home Sunshine Pharmaceutical Technology Co., Ltd
Novartis investigational lung cancer therapy capmatinib (INC280) granted FDA Breakthrough Therapy Designation for patients with MET-mutated advanced non-small cell lung cancer | Novartis

Osimertinib plus savolitinib in patients with EGFR mutation-positive, MET-amplified, non-small-cell lung cancer after progression on EGFR tyrosine kinase inhibitors: interim results from a multicentre, open-label, phase 1b study - The Lancet Oncology

A Phase 2 Study of Capmatinib in Patients With MET-Altered Lung Cancer Previously Treated With a MET Inhibitor - Journal of Thoracic Oncology

The promise of selective MET inhibitors in non-small cell lung cancer with MET exon 14 skipping - ScienceDirect