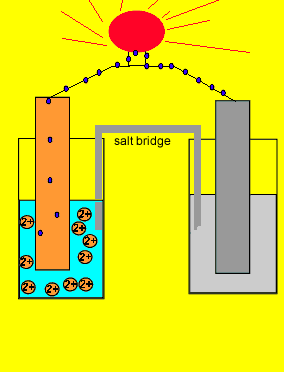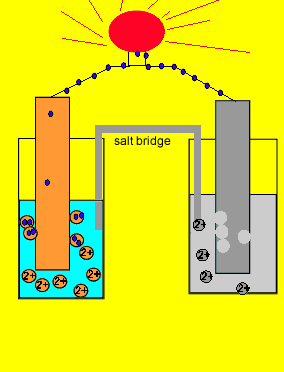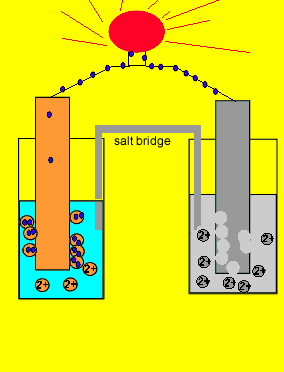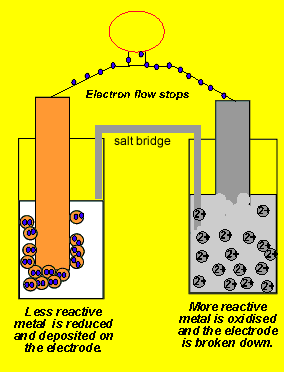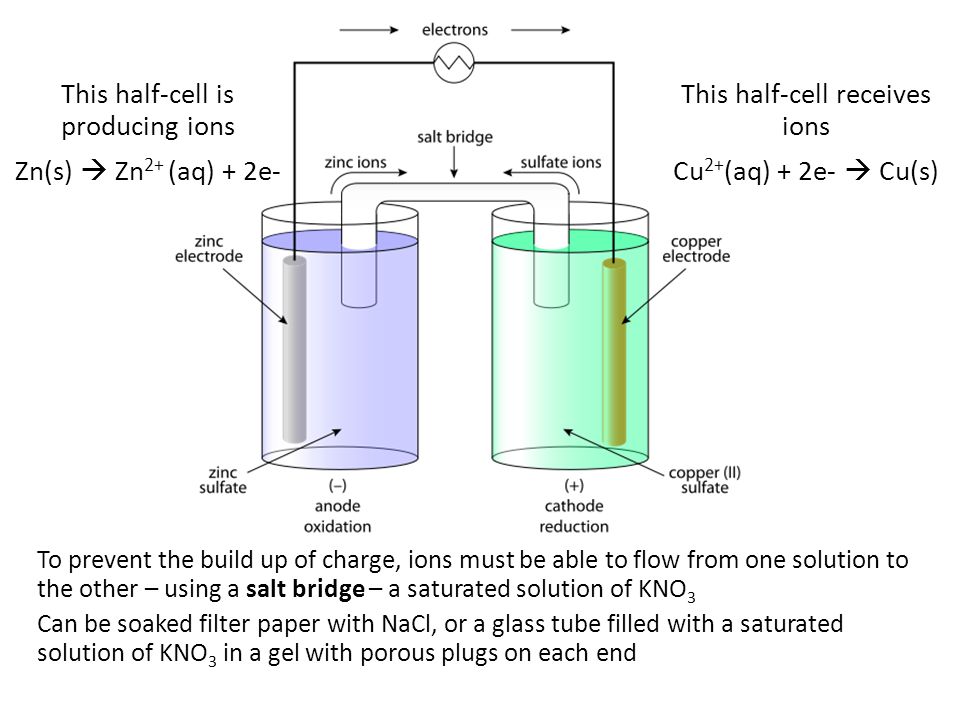
9.2 Electrochemical cells. Two types of electrochemical cells Voltaic cell Spontaneous Chemical Electrical Uses activity differences between two metals. - ppt download

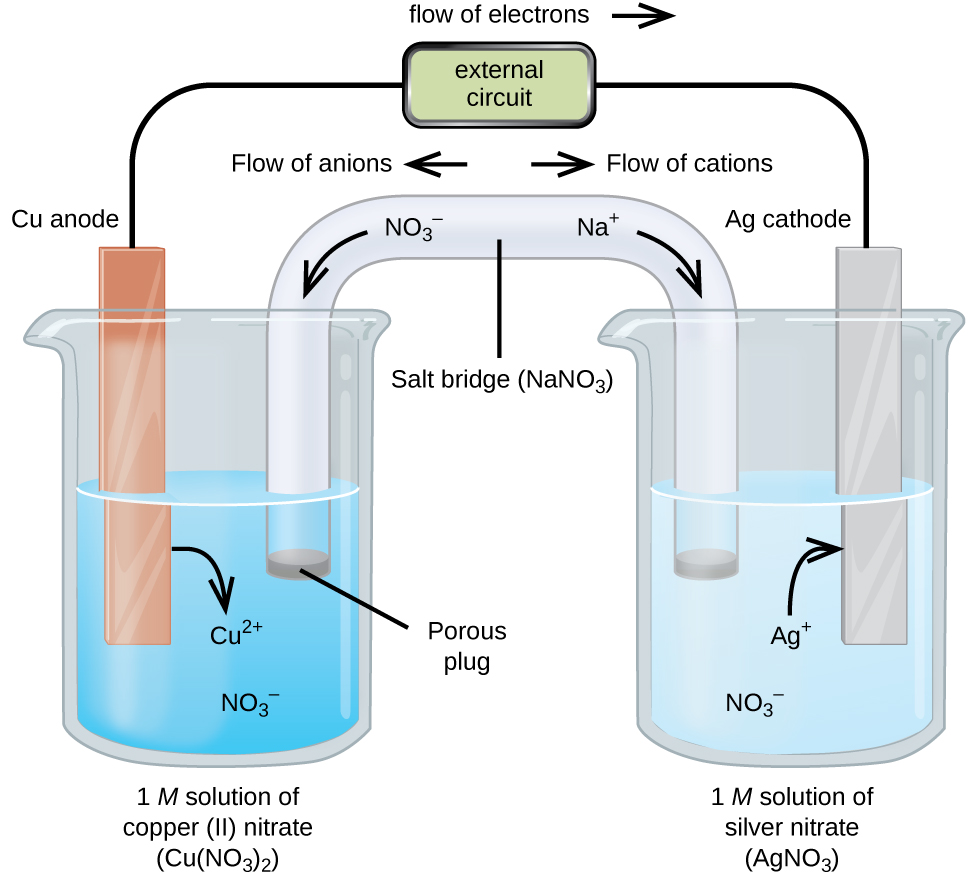
Filter Paper Salt Bridge in Potentiometric Titrations: An Undergraduate Laboratory Chemical Education Article

Filter Paper Salt Bridge in Potentiometric Titrations: An Undergraduate Laboratory Chemical Education Article



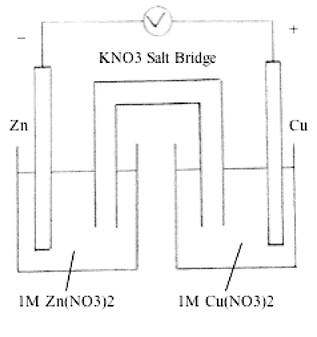

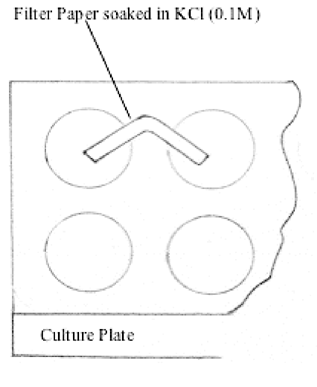


/saltbridge-5af43fcf875db900368d1853.jpg)
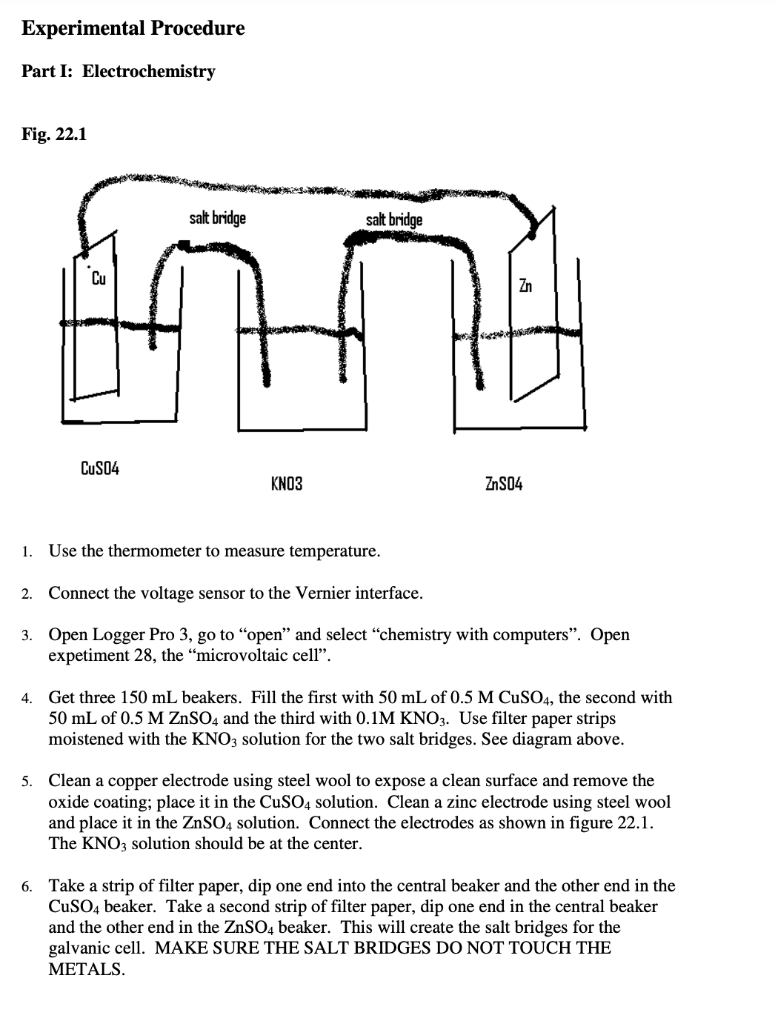

:max_bytes(150000):strip_icc()/GettyImages-10943484541-1145b5ddea574a1ab5d32aa398291feb.jpg)